
The deaths of the 66 children in Gambia may have been caused by contaminated cough and cold syrups, according to World Health Organization Director-General Tedros Adhanom Ghebreyesus.
The syrups were manufactured in India and distributed outside of West African countries, causing heart injuries in the babies.
According to the WHO, laboratory testing revealed unacceptable levels of potentially life-threatening contaminants, and the products may have been distributed beyond the West African country.

Late Thursday, India’s health ministry said it had been informed of the WHO’s findings last month and was awaiting the results of its lab tests on the four drugs.
It went on to say that the corporation was not licensed in India to dispense the four merchandise and had only produced and shipped them to The Gambia.
“It is customary for the importing country to test these imported products on quality parameters and satisfy itself as to the quality of the products,” according to the ministry statement.
Following the World Health Organisation’s health alert, Maiden Pharmaceuticals did not respond to AFP requests for comment.
Several times, Indian regulators have expressed interest in the company.
According to the FDA’s website, the company has received four warning letters this year for “substandard” product manufacturing based on batch tests.



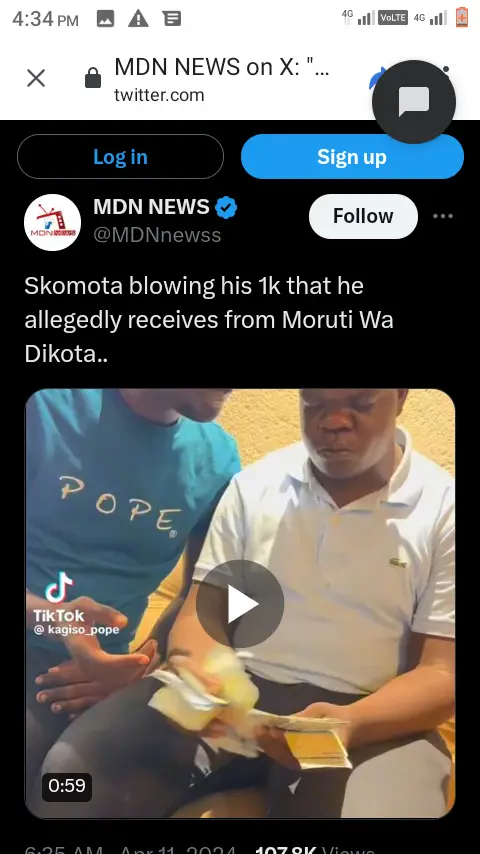






Awesome story hleee…Mara Sad hey🥺😢
Thank you for the feedback babe. This life thing has no balance